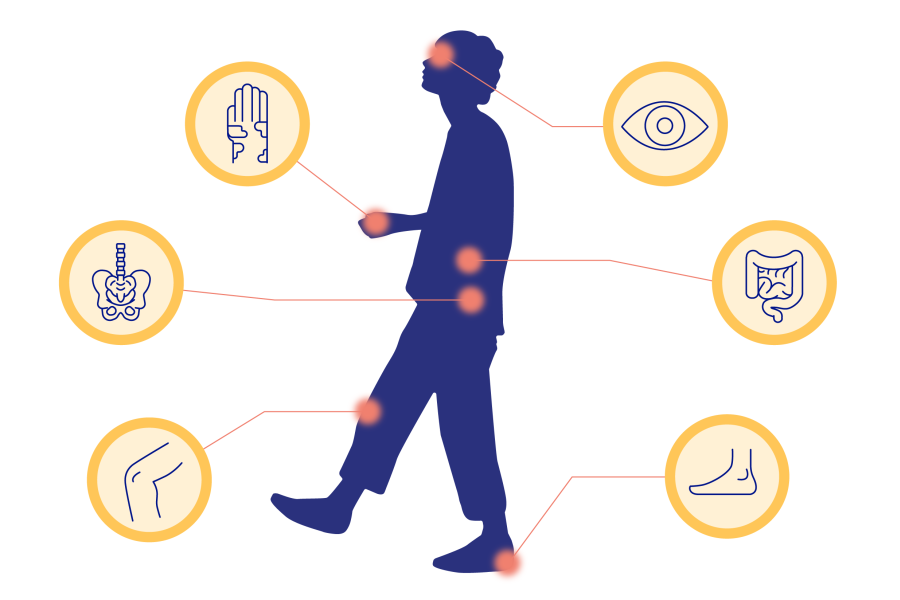
There are many clinically validated targets that drive pathogenic inflammation in spondyloarthritis, including IL-17, TNF, IL-12*, IL-23* and JAK-STAT.1–3
What are the cytokines implicated in SpA pathobiology?
Play the animation to accompany Assoc. Prof Philipp Sewerin in deciphering the role of extracellular cytokine targets in SpA pathobiology.
IL-17A and IL-17F are pivotal drivers of inflammation in PsA, axSpA, PSO and HS.4–7 They share overlapping biology,8–14 and can be produced by multiple lymphocyte subsets, a process which can be independent of IL-23.2,15,16
IL-17A and IL-17F:
Form homodimers and heterodimers9,10
Use the same receptor complex11,12
Are both expressed at sites of inflammation in PsA, axSpA, PSO, and HS4,7,13,14,17,18

Figure adapted from references8–14
However, IL-17A and IL-17F are also regulated and expressed differently:

Regulation
STAT5 induces preferential expression of IL-17F over IL-17A, whereas STAT3 enhances IL-17A over IL-17F20

Kinetics
Over time IL-17F becomes the dominant cytokine expressed by Th17 cells20†

Target Tissue Expression
IL-17F expression is high relative to IL-17A in tissues and in vitro models of PsA and axSpA20–22
†IL-17A is more potent than IL-17F in human skin cells, whilst IL-17F is more abundant than IL-17A in patients with psoriasis.21 CD4+ T cells stimulated with anti-CD3 and anti-CD28 in addition to brefeldin A between 0–72 hours.20
What disease manifestations are these cytokines implicated in?
Explore our resources below to find out the answers to this question and more today.
Join Prof Dennis McGonagle and Prof Lars Erik Kristensen in unraveling the role of the IL-17 family in the pathogenic inflammation and manifestations of SpA in...
PsA and axSpA have overlapping disease manifestations which are driven by inflammation.1 Explore several extracellular cytokines that are implicated in these...
Watch Prof Lars Erik Kristensen and Prof Frank Behrens discuss the role of the IL-17 family of cytokines in SpA pathobiology.
*Validated for PsA but failed for axSpA.1
EU-DC-2400159
Date of preparation: February 2025